Discussion
A brief talk about Pulmonary Embolisms
Pulmonary embolism (PE) is not well studied in the pediatric population. Although it is unusual in children, the incidence probably will increase due to improvements in pediatric care. In 1994, the Prospective Canadian Pediatric Registry showed that the incidence of PE was 0.86 per 10,000 pediatric hospital admissions annually1, and in 2001, the Prospective Dutch pediatric registry showed an incidence of 0.14 per 100,000 children annually2. The incidence is most likely underestimated due to the silent or non specific clinical presentation. The following are known risk factors for PE: Central venous lines, immobility, heart disease, ventriculo-atrial shunt, trauma, neoplasm, surgery, dehydration, shock, obesity, burns, nephrotic syndrome, sickle cell disease, medications (estrogens), and thrombophilic conditions. However, central venous line related PE is the mostly recognized mechanism in pediatrics.
We all are aware of the role of the ventilation/perfusion (V/Q) scan and CT angiography in diagnosing PE, but there are diagnostic tools which include the quantitative D-dimers test in the blood. Different techniques are used, but the most sensitive one is the ELISA technique. In adult studies, the sensitivity was up to 98%3. The cutoff point for a positive test is 500 ng/ml. The test is not specific to PE and it can be positive in other conditions such as DIC, surgery, trauma, tumor, systemic inflammatory disease, severe infection, sickle cell crisis, pregnancy, and after a myocardial infarct. Other useful tests include arterial blood gas (ABG) and pulmonary function. In the ABG, the PaO2 would be low, while the PaCO2 can be high, normal or low (depending on the timing of the patient's presentation). The A-a gradient could be high or low. PFT's may show a restrictive pattern (late in the course of the disease due to pulmonary infarction) and a decreased DLCO due to loss of alveolar volume available for gas exchange.
I will now talk about the clinical assessment of PE which means the frame work of diagnostic tests that influence the probability of PE, and it is divided into two categories: non standardized (empirical) and standardized clinical models (prediction rules). Empirical assessment basically includes 2 non-invasive imaging techniques that are the V/Q scan and CT angiography. In 1990, the "PIOPED" study (Results of the prospective investigation of pulmonary embolism diagnosis) tested the sensitivity and specificity of V/Q lung scan in patients with suspected PE4. The patients with a suspected PE had a V/Q scan and they were categorized into high, intermediate, or low probability scans. Patients then had a confirmatory angiography test. The PE was confirmed by angiography in 88% of high probability scans, and 8% or less of normal or near normal scans. The study indicated that a high probability scan is sufficient evidence to treat for PE and that therapy can be safely withheld in normal or near normal scans. However, forty four percent of the study population had intermediate probability scans, and PE prevalence was 33% in those patients. In 2006, PIOPED-II study tested the spiral CT angiography (CTA)5. It showed that CTA had sensitivity of 83%, and specificity of 96%. If CTA was combined with CT venography of the pelvic and thigh veins the sensitivity increased to 90%, and specificity to 95%.
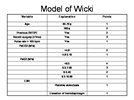
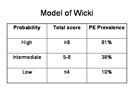
In the standardized clinical assessment there are 2 clinical models: model of Wells and model of Wicki6,7. These models were designed to help to decide about the management plan in adult outpatients. They have not been validated for pediatrics.
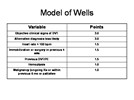
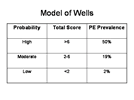
In their model, Wells et al used the medical history and physical examination to divide the patients into high, intermediate, and low clinical probability based on the total score.
The model of Wicki used medical history, physical examination, laboratory investigations, and medical imaging to get the total score.
In 2006, the investigators of PIOPED-II study recommended to add quantitative D-dimer test, using the ELISA technique to the work-up, and combine the standardized and empirical assessments into different pathways when investigating patients with suspected PE8.
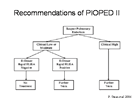
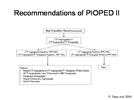
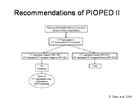
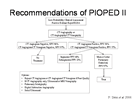
Any patient with suspected PE would be placed into a clinical category, based on the model of Wells or Wicki. If the patient has a low or moderate probability, then D-dimer testing should be done. If the result is below the cut-off point then no more investigations nor treatment are warranted. However, if it is positive, then the patient needs imaging studies. If the patient falls into the high probability category, then he/she needs CTA +/- CT venography. If this is positive then treat the patient, but if they are negative then there are different options that you can choose from (figure 19). If the patient has a moderate probability, has positive D-dimer test, and positive CTA then he/she should be treated. However, if the CTA +/- CT venography is negative then you may not treat or choose from the provided options (figure 20). Lastly, if the patient has a low probability, has positive D-dimer test but negative CTA, then do not treat. However, if the CTA is positive and showing main or lobar PE then treat (figure 21).
Mortality rates in the Canadian and Dutch registry of venous thromboemboli were 10%. In the Canadian Childhood Thrombophilia Registry of catheter-related VT, the mortality rate was 18%9. In children, the recurrence rate and long-term effects on pulmonary function are unknown.
Group discussion and questions
Q: The pleural effusion was a mixture of transudate and exudate type, so what is the mechanism of the effusion in pulmonary emboli?
A: The pleural effusion in pulmonary embolism is exudative and it is due to necrosis of the pulmonary parynchema with release of inflammatory mediators that alters the capillary permeability and result in the fluid collection in the intrapleural space. However, the oncotic pressure would rise in the tissue and it will drag more fluid in the space. So the pleural fluid may have a mixed picture of both transudative and exudative type.
Q: The data that was presented about the PIOPED pathways for the work-up and treatment of pulmonary embolism, and the D-dimer test, is that applicable to the pediatric population?
A: No. All above data from studies that was conducted on adult patients. They were not intended for pediatric patients and we can not extrapolate their results and apply them to the pediatric patients with suspected PE. Up to now, I am not aware of any medical literature that studied those tests in pediatrics. That is why at Montreal Children's Hospital, we investigate any patient with suspected PE through the empirical-assessment approach, i.e. V/Q scan or CTA +/- venography of the pelvic and thigh vessels. We also send for the D-dimers before the imaging investigations. If they are positive then it is supportive evidence, but if they are negative and we still believe that the patient has a good chance to have PE, we proceed to the CTA.
Comment
Although this case might be easy and obvious for an adult respirologist or even internist, the case is different in pediatrics. As a pediatric pulmonary fellow who finished 4 years of pediatric residency, I know that it is very hard for the pediatricians to include PE in their differential diagnosis for chest pain or pleural effusion. Even during my presentation, the pediatric respirology fellows did not include PE in their differential diagnosis of the pleural effusion. I think this is because of the paucity of this diagnosis in the pediatric population, and the non specific presentation and clinical signs of PE, in particular if the patient has no obvious risk factors. I think that PE is still a diagnosis of exclusion in pediatrics, especially if there are no obvious risk factors. However, the pediatricians and in particular pediatric respirologists should have a high index of suspicion.
