i)
Treatment
1.
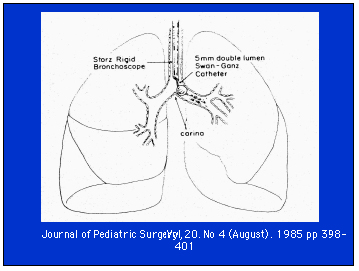
Whole lung lavage is the treatment of choice, first described by
Ramirez in 1965
–
Tolerated because rapid alveolar fluid clearance (53%
within the first hour) Chestnutt et al
–
Improved FVC, TLC, PaO2 at rest and with exercise
–
Improved DLCO, A-a gradient and shunt
–
Other reported: decreased LDH, gradual radiographic clearing,
improved V/Q
–
Complications: long anaesthetic time, hypoxemia, hydrothorax,
bronchoconstriction, pneumonia, and electrolyte disturbances, complications
of cardiopulmonary bypass if it is utilized.
–
Outcome: 60% good response with 2 lavages per lung, <
10% non-responders (Shah, 2000, Thorax)
–
It is unknown what differentiates those that responded
to WLL and those that did not.
When
and how to proceed with WLL?
WLL is indicated when respiratory symptoms impair the quality of life
or lung function deteriorates (Claypool et al, 1984, Chest). Kariman
et al, (1984, Chest) suggested proceeding with WLL if n =23 (lavage
if Pa02 < 70 mmHg, or P(A-a) > 40 mmHg) based on sample size of 23 patients.
More severely lung lavaged first, if both affected (L) lavaged first
because right has larger volume and better ventilatory capacity, second
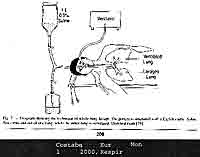
lung lavaged 3-7 d later (Costabel, 2000, Eur Resp Mon)
Youngest
reported to have WLL (2 kg) without ECMO (Moazam et al, 1985, J Peds
Surg)
–
Manual percussion during the instillation and evacuation
improves qualitative yield (better optical density of removed fluid)
(Hammon, 1993, Chest)
–
Manual ventilation for 20 tidal breaths during second
half of drainage procedure, allowed better drainage (Bingisser, 1998,
Chest)
–
Other variations: Using ECMO:
Adults (n=5) unilateral lung lavage;
One case – Bilateral sequential whole lung lavage with venovenous ECMO (Cohen, 2001, Chest), other reports of adults having sequential lavages
Children:
(n=3) bilateral whole lung lavage with ECMO (Mahut et al, 1992, Peds Pulm) – Liquid ventilation (one case report in an infant) (Tsai et al, 1998, Peds Pulm). – Hyperbaric oxygen (Jansen et al, 1987, Chest)
Trypsin - segmental lavage - adults (Nagasaka et al, 1996, Thorax)
2.
GM-CSF:
–
Naturally occurring molecule
–
Hematopoetic effects on multiple cell lines, stimulates
proliferation and differentiation of multiple cells: white blood cells
–
Side effects: Mild 20-30%: local reactions, fever, myalgias,
headaches, flu-like. Severe <2%: Anaphylaxis, cardiac failure, leaky
capillary syndrome.
–
Daily subcutaneous injections, dose: 5-10 ug/kd/d iv/sq
–
Expensive
–
Case reports of treatment with GM-CSF in pulmonary alveolar
proteinosis in adults: - reasonable efficacy (n=1) (Seymour, 1996, NEJM)
–
1/3 responded (Schoch, 1998, Eur Respir Journal)
–
1/1 responded (Barraclough, 2001, Thorax)
–
Clinical trials: small n
–
open label, 3 patients out of 4 total responded (Kavuru,
2000, AJRCCM)
–
4/8 responded (Thomassen, 2000, Clinical immunology)
–
6/14 responded (Seymour, 2001, AJRCCM)
–
better response in older, longer time since dx, normal
LDH, higher SP-B level
–
Limited experience in children in this setting.
j
) PAP - Prognosis in Children
The following page provides data on the reported cases. The authors
have not been contacted for further information. All the information
available in the articles is presented. Infants appear to have a worse
prognosis. Spontaneous resolution has been reported in children, but
the details of these children are not known , nor the factors that make
spontaneous resolution more likely.
SUMMARY
– PAP and SP-B very different diseases– SP-B deficiency: poor prognosis without lung transplant, refractory to treatment, role of gene therapy in the future
– PAP: variable outcome, younger patients have a poorer prognosis
– whole lung lavage is the treatment of choice
– ? role of GM-CSF experimental
– reported reoccurrence after lung transplant
THIS CASE IN RETROSPECT
With this patient HRCT may not have helped in making the diagnosis of PAP as the findings were not classical. We are considering the possibility of starting GM-CSF therapy for this patient.Discussions raised during the case presentation were: GM-CSF is being used as inhalational therapy on an experimental basis and perhaps this is the route to pursue given the lack of improvement with lung lavage. This would direct the treatment to the site of the disease and avoid systemic toxicity and side effects.
The possibility of lung transplant was also raised since that may lead to cure of PAP. It is uncertain whether this patient has a congenital heterozygous form of surfactant deficiency given her extensive neonatal problems or whether this is truly acquired PAP. However, one has to weigh the risks and benefits of lung transplantation in the context of her current disease and quality of life. In summary, case highlights some specific clinical issues surrounding the diagnosis and treatment of pulmonary alveolar proteinosis.
Special thanks to Dr. Alfred Pinto for the pathology slides.
THANK YOU FOR JOINING US FOR THE CROSS-CANADA PAEDIATRIC RESPIRATORY ROUNDS. HOPE TO SEE YOU NEXT TIME.
Thank you to AstraZeneca for their support of this program and website.